project
Fluorous Chemistry
Highly fluorinated organic compounds exhibit unique physical properties that allow them to be both immiscible in common organic solvents and also water. The density of these compounds is higher than those of typical organic compounds. We are developing novel synthetic methodology and new functionalized materials that capitalize on the specialized properties of fluorous compounds.
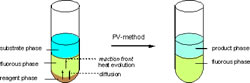
The phase-vanishing method
The phase-vanishing (PV) method is based on spontaneous reaction control by diffusion of reagents into fluorous media, such as perfluorohexanes (C6F14). The original PV method is a triphasic reaction system in a test tube composed of a bottom layer: reagent phase, middle layer: fluorous phase, and top layer: substrate phase. The fluorous phase acts as a liquid membrane to transport the reagent from the bottom layer to the top organic phase which contains the substrates. Upon completion of the reaction, the bottom layer disappears and two phases remain. We have applied the PV method successfully to the bromination of alkenes with Br2, halogenation of alcohols with SOBr2 and PBr3, and also demethylation of methoxyarenes with BBr3, cyclopropanation of alkenes by CH2I2-AlEt3, and Friedel-Crafts acylation of aromatic compounds with SnCl4. Recent advances include (1) a “quadraphasic” PV method, in which an aqueous “scavenger” phase was added to the original triphasic PV method to remove acidic by-products, and (2) a photo-irradiative PV method in which the PV bromination method is successfully combined with photo-irradiation, providing a useful tool for generation of HBr in the fluorous phase. The HBr generated mat then undergo further reaction for examples the addition to an alkene. This allows for two distinct reactions to occur in “one pot”.
Fluorous reagents and solvents
A fluorous ether, termed F-626, is a cosmetic ingredient made by Kao corporation. It has been demonstrated that this fluorous ether is an easily recycle alternative to high-boiling point organic solvents such as o-dichlorobenzene or diethylene glycol. Fluorous DMF and fluorous LDA have been prepared and it has been demonstrated that they can be used as recyclable solvents or reagents.
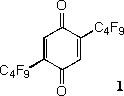
Utilizing the electron-withdrawing effects of fluorous ponytail.
Perfluorinated alkyl chains (termed “fluorous ponytail”) are usually attached to the core of catalysts or reagents with an alkyl spacer when fluorous catalysts or reagents are prepared. The spacer, which is normally a dimethylene or trimethylene group, suppresses strong electron-withdrawing effect of a perfluorinated alkyl group. We are interested in the electronic effects of fluorous ponytails and synthesizing fluorous compounds in which perfluorinated alkyl group are attached to the core of catalyst or reagent directly. For example, fluorous benzoquinone 1 was prepared as a recyclable strong oxidizer.
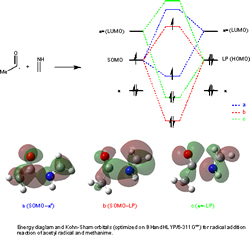
Molecular Modeling
Computational investigations into mechanistic details of chemical processes are carried out. We are especially interested in reactions involving an acyl radical. For example, acyl radicals are known to attack at the nitrogen end of an imine (C=N) bond exclusively while similar reactions with alkyl radicals beset by poor regioselectivity. We have shown clearly the reason using DFT and NBO analysis (see below). Many calculation and experimental joint projects have been concluded in collaboration with groups both domestically and overseas. Collaborators includes Prof. Ilhyong Ryu, Osaka Prefecture University, Japan; Prof. Carl H. Schiesser, the University of Melbourne, Australia; Prof. Shigeo Hayakawa, Osaka Prefecture University, Japan; Prof. David J Procter, Manchester University, UK. This fundamental radical chemistry provides vital mechanistic insight into important radical processes.